The evolution of computerized systems for the production of dental restorations associated with the development of new ceramic material microstructures has caused a significant change in the clinical workflow for dentists and technicians as well as the treatment options offered to patients. One of the most important changes in this scenario has been the introduction of monolithic restorations made from high-strength ceramics, such as zirconia.
This concept per se is not new, as ceramic materials have been used for the production of monolithic restorations for a relatively long time, but it was only when zirconia began to be used to produce full-contour crowns that dentists and technicians became more confident in indicating a ceramic material for crowns and bridges in the subsequent area. In fact, by offering monolithic prostheses, clinicians are able to overcome one of the major problems associated with multilayered restorations, which is the fracture of the low-strength veneering layer , usually made from a feldspathic dental ceramic. However, certain health issues can occur by utilizing a monolithic zirconia repair and ought to be taken care of, such as wearing antagonist dentition and maintaining the natural dentition 's cosmetic characteristics .
Classification of ceramics
Dental ceramics can be categorized
in a number of ways, including form, manufacturing procedure, temperature
fusion, microstructure, translucency, fracture resistance and abrasivity.
Classification by Composition
Ceramics can be classified into
three groups by composition2: ceramics that compose primarily of glass, those
composed of particle-filed glass, and polycrystalline ones. Ceramics which
consist mainly of glass have the highest esthetics. Often manufacturers add
small amounts of filer particles to monitor the optical effects which mimic
natural enamel and dentin. Generally speaking, the more filer particles added
to a ceramic, the greater the increase in the mechanical properties but the
greater the decrease in the esthetic characteristics. Polycrystalline ceramics
contain absolutely no glass. These aren't porcelain as noted earlier.[2]
Classification by Processing Method
Another approach to the
description of ceramics is by the method of manufacturing them. This includes
the production of powder / liquid, slip casting, hot ceramic pressing, and
computer-aided additive and subtractive design / computer-aided manufacturing
(CAd / CAM).
Classification by Fusing Temperature
Dental porcelain is defined by the
temperatures of its fiing. These groups are defined as high-fusing (1300 ° C),
medium-fusing (1101 ° C to 1,300 ° C), low-fusing (850 ° C to 1,100 ° C), and
ultra-low-fusing (< 850 ° C). 7 Denture teeth are a case in point of high
fusion porcelain. Depending on the system, crown and bridge porcelain may be
medium- or low-fusing, and ultra-low-fusing porcelain would be used for
porcelain and glazing. Some now apply to just two categories — high- or
low-fusing porcelain — with a distinction defined at 800 ° C to make it less
difficult. [3]
Classification by Microstructure
As mentioned earlier, porcelain
has two different phases: the phase of glass (responsible for esthetics) and
the phase of crystalline (related to mechanical strength). In the case of
feldspathic porcelain, when feldspar is melted, crystalline mineral called
leucite (potassium-aluminum-silicate) forms. Feldspar undergoes incongruous
melting between 1.150 ° C and 1.530 ° C to form leucite crystals. Incongruous
melting is a process in which one material does not evenly melt and forms a
different material.7 The leucite crystalline phase has a diffaction index
similar to the glass matrix which, in this case, contributes to the overall
esthetics of porcelain.17The leucite content of porcelain is associated with
the propagation resistance of cracks. Greater content of leucite means a
greater decrease in the propagation of a crack.18 This type of porcelain is
called leucite-reinforced. Microporosities are formed on the surface during the
sintering process of all-ceramic restorations which lead to crack initiation
and propagation and ultimately result in failure. [4]
Classification by Fracture resistance
A quantitative means of describing
the tolerance of a ceramic to brittle fracture when a crack is present is
called the "fracture toughness," which is the capacity to tolerate
crack growth.1 When a substance has a high value of fracture toughness, it is
likely to undergo ductile fracture. Brittle fracture is very characteristic of
materials with a low fracture toughness value.48 Flexural strength (rupture
module or bend strength module) is defied as the ability of a material to
withstand deformation under load. Flexural strength reflects the maximum stress
encountered in the material at the time of breakup and is measured in terms of
stress.7 For example, the estimated flxural strength values for zirconia vary
from 900 MPa to 1,100 MPa,49,50 and fracture was estimated from 8 MPa to 10
MPa. [5]
Classification by Abrasiveness
Ceramic restorations were known to
induce opposed enamel wear. 53 The abrasiveness of a dental ceramic is
determined primarily by the smoothness of the material. 54 For wear to occur,
friction must evolve by mechanical interlocking of the two wear bodies. Low-fusing
porcelain was developed with the idea of reducing the abrasiveness of the
ceramic surface to incorporate fier-sized leucite particles in lower
concentrations. [6]
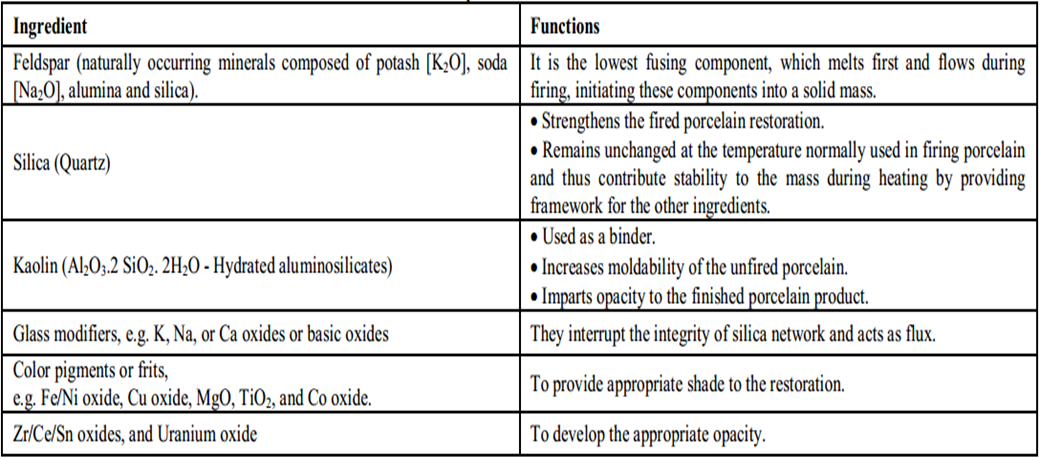
Composition
Dental ceramics are composed primarily of crystalline minerals and matrix
glass. Crystalline minerals include feldspar, quartz and alumina and maybe
kaolin as a matrix of glass. Table 1 discussed the detailed composition of the
dental ceramics [7]
Zirconia phases
Zirconia (ZrO2) also occurs in
several phases: monoclinical, tetragonal, and cubic. Zirconia is monoclinic
until 1170 ° C. It then converts to tetragonal until a temperature of 2370 ° C
reaches. At this temperature, zirconia becomes cubic until the melting point is
at 2680 ° C. (Figure 1)shows those major zirconia polymorphs. Phase transitions
of Zirconia are reversed after cooling and followed by volume expansions. With
a change of the theoretical density from 6.06 to 6.1, cubic zirconia transforms
under 2370 ° C into tetragonal. The tetragonal phase transforms to monoclinic
at around 1170 ° C, and the density decreases from 6.1 to 5.83. The
transformations into the monoclinical phase are obviously accompanied by volume
expansions of approximately 4%. The transformed grains undergo volume-induced
strains and changes of shape induced by phase transformation, accompanied by
strains of shear. These strains apply stress to grains adjacent to them. Thus
sintered pure zirconia suffers a microscopic cracking, which limits its use as
refractory ceramics or pigments. This condition improved in 1929, when zirconia
stability was first achieved.
Additives to zirconia such as
calcium oxide, magnesium oxide, cerium oxide and yttrium oxide are used to
stabilize high-temperature phases and are given the name "Partially
Stabilized Zirconia (PSZ)." At room temperature, PSZ as a biomaterial
contains the cubic phase as the major phase and the monoclinic phase as the
minor phase. Another minor phase of PSZ is the metastable tetragonal phase,
which exists by a quantity that depends on many "material design
parameters" such as stabilizer quantity, zirconia and stabiliser particle
sizes, chemical history, and processing method. The processing method affects
the homogeneity of the end powder mix and the final shape of the particle,
which affects the surface area. The residual stresses in the bulk are dependent
on the cooling procedure for the sintered product, and the more likely the
required phase transformation will take place by decreasing it.
The tetragonal metastable phase
helps enhance the toughness of the fracture through a mechanism called
toughening transformation. It is magnified at the crack tip as a force is
applied, which is called force concentration. The magnified stress will exceed
a value necessary to establish transformation of the tetragonal – monoclinic
process. This is combined with an extension of length, and would apply a
compression on the edge of the crack. As a result, crack growth is retarded and
the resilience of the cracks is decreased. The removal of some of the crack
energy that is caused by phase transition also leads to retarding the crack and
improving the resilience of the fracture. Obviously, the increased toughness of
the fracture improves the product 's reliability and increases its service life
[8].
Stabilization of zirconia
Zirconia (ZrO2) has three
polymorphic forms: tetragonal, monoclinical and cubic. At room temperature, the
monoclinic phase is stable; the tetragonal phase is a metastable process
developing at 1170–2370°C; and the cubic process starts to develop above
2670°C. The cubic – tetragonal transition takes place on cooling from high
temperature with a small expansion of the volume of the unit cell. In the other
hand, high-volume expansions (3 percent – 5 percent) follow the
tetragonal–monoclinic and cubic–monoclinic transitions. Therefore the sintered
pure zirconia undergoes the development of cracks during the cooling process
caused by significant stresses. To overcome this difficulty, oxide additives,
such as CaO, MgO, and Y2O3, are used when cooled from the sintering temperature
to room temperature to stabilize the cubic or tetragonal phase of zirconium. So
the powder of zirconia is called stabilized zirconia.
The addition of approximately 8
mol percent CaO or MgO creates partly stabilized zirconia (PSZ), i.e. during
cooling, the cubic or tetragonal transition to monoclinical phase is bounded,
and thus crack forming does not occur. Roughly speaking, the monoclinic phase
mount is about 10 percent -20 percent and can be lowered by thermal ageing to
less than 5 percent. With PSZ, improved mechanical properties are obtained due
to the toughening transformation (TT) phenomenon. TT is an induced transition
of the tetragonal phase into the monoclinical phase that is exacerbated by
access to crack energy from stress. Within several microns of the crack tip,
the grains which make up the microstructure turn into monoclinic. The volume
expansion is followed by compressive stress which prevents the propagation of
cracks.
Completely yttria-stabilized
zirconia (Y-TZP) with about 8 mol percent Y2O3 can be achieved. Because of the
increased homogeneity of the powders and the smaller particle size of the
zirconia and Y2O3 additive, 5 and 3 mol percent of Y2O3 was adequate for
complete stabilisation with the advancement of powder technologies. Currently,
an addition of 3 mol percent of Y2O3 to produce stabilized tetragonal zirconia
powder, or polycristals (3Y-TZP), is commercially available as nanopowder and
is often used in the production of sintered zirconia.
Zirconia is often produced with
the help of decomposition agents via the decomposition of zircon (ZrSiO4) and
followed by a purification phase. Specifics of the process depend on the
existence and desired properties of the raw materials. Low-temperature
high-purity zirconia tetragonal can be produced using a special technique
called the sol – gel process. The sol – gel cycle usually involves acid
dissolving a salt of zirconium accompanied by nucleation and development of
zirconia. So the sol – gel technique is used to produce nanosized zirconia by
controlling the growth of the particles. Other chemical techniques are
available for preparing zirconia, but are not covered in this book. One of the
benefits of the sol – gel process is that it can integrate additives, such as
Y2O3, during the process. Therefore a high degree of homogeneity and particle
size regulation is achieved which helps to stabilize tetragonal zirconia.
In the mid-1980s MgO-PSZ was
introduced as a bioceramic for the production of ball heads to replace total
hips. Better wear resistance, strength of bending, and toughness of fracture
were obtained compared with alumina. By comparison, the large grain size of the
sintered substance (40–50 μm) increases the wear rate of the acetabular portion
of polyethylene. Thus yttria-stabilized tetragonal zirconia ball heads were
added, as the sintered product's submicron grain size greatly improves wear
properties.
The manufacturing process of
yttria-stabilized tetragonal zirconia involves a precise and complicated heat
treatment program which ensures precipitation of the tetragonal acicular phase
at the cubic phase's grain boundaries. The durability of zirconia was found to
be highly dependent on these processing conditions. Furthermore, the
environmental conditions the implant faces also affect zirconia stability.
Explicitly, the zirconia implant surface is exposed to the implant 's body
(synovial) fluid, which causes implant degradation at low temperatures (LTD).
LTD is the monoclinic phase increase that contributes dramatically to the
ruggedness improvement. The LTD impact can be minimized by over-stabilizing the
tetragonal process by a small rise in the stabiliser. In the other hand,
improved stability does not impede the TT and the mechanical properties should
be that accordingly. These complexities pose a minor downside for the
operation. The production of zirconia balls has since been slowly reduced [8,
9].
Properties of zirconia
An excess of raw materials such as
zirconia minerals (ZrSiO4) and baddeleyite (β-ZrO2) can be found in nature for
the manufacture of zirconia (about 0.02 percent of the earth's crust). Big
zirconia deposits are present in Brazil as baddeleyte while they can be found
in Australia and India as ZrSiO4.
Zirconium is a transition metal
element, and as a white and ductile metal it has a pure crystalline shape and
as a blue-black powder it has an amorphous form. Even as expanding, it is the
18th element in the earth 's crust, zirconium can be present in nature either
in association with silicate oxides or as free oxide. Zirconia takes place in
three phases: monoclinical (m), cubic (c), and tetragonal (t). The monoclinic
phase is stable up to 1170 ° C in pure ZrO2; but the transformation on cooling
appears under 1170 ° C at 100 ° C. When it cools down it has 3-4 per cent
volumetric expansion. The cracks may appear as this change in volume is enough
to exceed the ZrO2 elastic limit. Passerini and Ruff et al., quoted by Lughi V,
found that by alloying it with other cubic oxides, called stabilizers, zirconia
can remain stable at room temperatures. Until now, the most widely used
stabilisers for the application of biomaterials are CaO, MgO, Y2O3 and CeO2,
but only ZrO2-Y2O3 has a norm of self-ISO for surgical use[10].
Biocompatibility
No local or systemic cytotoxic
symptoms or allergic reactions have been observed after detailed
biocompatibility tests of the zirconia. It has been shown acceptable for the
bone response of zirconia in vivo and for the inflammation adjacent to
zirconia. Additionally, bacteria and pathogen appear to adhere just as much to
zirconia as other materials do[11].
Optical characteristics
The most important components of
presentation of an esthetic tooth are: light, fluorescence, opalescence and
translucency. One big downside of restorations of complete contour zirconia is
its opacity[12].
Translucency
Colored zirconia with improved
translucency has been developed recently to precisely match human teeth
colours. The new material's flexural strength is 900-1400 MPa, and has a crack
tolerance of up to 6 MPam 1/2. Such conveniences have made it widely used by
the Zirconia for inlateral applications of crowns and bridges. It is necessary
to replicate the translucency of the natural tooth for excellent esthetics,
since it provides an enchanting natural appearance of the prothetics.
Translucency is the property of the surface which allows the passage of light
and its dispersion, and then the objects are not clearly seen through the
material. This property could be defined as a state between total obscurity and
transparency. Translucency can be controlled by monitoring light absorption,
reflection, and propagation through the material. The translucency is higher
when there is low resistance and fast transmission. It has been stated in a few
studies that translucency is influenced by the thickness of the layer and by
the grain size [13].
X-ray opacity
Dental restorative materials have
varying degrees of opacity and provides useful diagnostic information. The
zirconia may be added in composite materials for dental filling because it
represents an opaque X-ray agent. In a research conducted to assess zirconia's
X-ray hardness, four different materials (pure titanium, NANOZR, Y-TZP, alumina
plates) were measured against an X-ray agent with the same thickness (0,2-2 mm)
and human tooth. Y-TZP and NANOZR have been found to exhibit increased
opacity[14].
Wear behavior
It is important to determine the
wear behaviour of monolithic zirconia restorations, but the wear of the enamel
of natural antagonists teeth in comparison to the zirconia material is of
greater clinical importance[15].
A natural occurrence of human
dentition is gradual wearing of teeth. Many factors contribute to the dental
wear such as diet, bruxism, strength of the oral muscles, thickness and
toughness of enamel, pH and quality of saliva and dental content. Each of these
variables have specific wear habits that often change the wear cycle. If we use
restorative fabrics with specific wear rates, the physiological wear is changed
[16].
Processing methods
CAD / CAM components used in
dental restorations are typically totally or partly sintered blocks and disks.
The state of those blocks and disks differentiates dental restoration
manufacturing.
With two different methods, dental
prosthetic restorations made from zirconia can be achieved using the CAD-CAM
technology [17].
In the first technique, the prothetic
restorations are milled from already sintered zirconia blocks, which means that
there is no shrinkage to the final structure, but there are certain
disadvantages, such as the reduced lifespan of the burs, due to their high wear
and the numerous flaws that occur during the machining process, which can
diminish the mechanical properties of the final prothesis. In this process, at
temperatures below 15000C, the Y-TZP blocks are subjected to a first
sinterization, increasing their density. Then, in an inert gas atmosphere, the
blocks are subjected to high pressure at the same temperatures, which allows
the obtaining of a very high density of more than 99 % of the theoretical one
[18].
In the second method, the zirconia
prothesis is milled from a block, replicating the final prothesis shape but
with larger dimensions, thus compensating for the shrinkage after sintering.
The structure of an oversized
zirconia (Zirkonzahn Ice Zirkon Translucent) after milling is shown in Figure
2.
The ceramics are then fired and
the structure contracts to the final level. After a cold pressing process,
which compacts the zirconia powders, the non-sintered zirconia blanks lead.
Through this way we obtain a very low pore size and a strong part distribution
inside the blank . The next step is the machining of various metal solutions
(cerium, bismuth, iron or a combination of them) by immersion and the coloring
of the restorations. As it passes through the last phase of sintering the color
develops. The concentration of the solution has a direct influence on the final
shade. A satisfactory coloring can be achieved by concentrations as low as
0.01mol percent. For a good result, we must follow the instructions of the
manufacturer as the final sintering temperature influences the color obtained.
At the end of the sintering process, the zirconia frame acquires its final
mechanical properties when it undergoes a contraction of about 25 per cent,
which means it returns to its correct dimensions. It is imperative to know the
exact volume shrinkage information for each blank block of zirconia to optimize
the fit of the restore. The vast majority of blocks have barcodes which give
the computer information about the density of the milling block and so we can
over-size the frame appropriately [19].
Milled frame of bulky zirconia,
separated from the blank, can be seen in Figure 3. In the final stages,
sintering is mandatory to remove any tension caused by the operation of surface
milling and to obtain the required density.
A multilayer coating technique is
used to veneer zirconia frame with compatible ceramics to create an aesthetic
look of the zirconia ceramic reconstruction. Figure 4 displays the layers of
Zirkonzahn Ice Zirkon Ceramics added (Ceramic Dentine A1, Ceramic Enamel S1,
Ceramic Transpa Neutral)

Uses of zirconia
Although there are currently many
types of zirconia-containing ceramic systems available, so far only three are
used in dentistry. These are polycrystals of yttrium cation doped tetragonal
zirconia (3Y-TZP), partially stabilized magnesium cation doped zirconia
(Mg-PSZ) and zirconiatoughened alumina (ZTA)
Dental posts located in Zirconia
Development of new post materials
(Figure 6) has begun the requirement for more esthetic posts, especially under
all ceramic restorations. In situations where all-ceramic restorations are used
to restore anterior teeth, metal posts can lead to unfavorable esthetic
results, such as a gray discoloration of translucent all-ceramic crowns and the
gingival margin around them. In addition, prefabricated corrosive reactions can
cause complications involving the surrounding tissues and oral environment,
including metal taste, oral burning, sensitization, oral pain, and other
reactions. These issues led to white or translucent posts made of zirconia and
other ceramic materials being created [20].
Crown and Bridge, Zirconia
Often used was the manufacture of zirconia structures for crown and bridge of either presintered or strongly isostatic pressed zirconia, as seen in (Figure 7). Zirconia systems deliver new insights in metal-free partial fixed dentures and single tooth reconstructions due to the high flexural power of more than 900 MPa of zirconium and showed positive initial clinical results[21].
Implant abutments located in Zirconia
This allowed clinicians to expand
their demand for implant-supported restorations as a result of using the
zirconia ceramics for the manufacture of tooth-supported restorations (Figure
8). The use of zirconia as implant-supported restorations is due to the greater
durability and lower zirconia elasticity modulus. Zirconia has some benefits
over alumina in stable and converted ways in order to overcome the issue of
alumina fragility and the consequent possible implant failure. These abutments
are distinguished by their color matching the tooth, their good compatibility
with the tissue and their lower accumulation of plaques[22].
Esthetic orthodontic brackets, based in Zirconia
In addition to the previously
mentioned dental applications, zirconia was also used for the manufacture of
esthetic orthodontic brackets. Polycrystalline zirconia brackets have been
offered as an alternative to alumina ceramic brackets, which are reported to
have the greatest toughness among all ceramics. They are cheaper than the
ceramic brackets of monocrystalline alumina but they are very reflective and
can display intrinsic colors making them less attractive. For both stainless
steel and nickel-titanium archwires, strong sliding properties were identified
along with decreased plaque adhesion, clinically appropriate bond strengths and
loci of bond failure at the bracket / adhesive interface. Keith et al.,
however, found no significant advantage of zirconia brackets over
polycrystalline alumina brackets in terms of their frictional properties[23].
References
1- Schley
JS, Heussen N, Reich S, Fischer J, Haselhuhn K, Wolfart S. Survival probability
of zirconia‐based fixed dental prostheses up to 5 yr: a systematic review of
the literature. Eur J Oral Sci. 2010;118(5):443-50.
2- Kelly
Jr. dental ceramics: what is this stuf anyway? J Am Dent Assoc.
2008;139(suppl):s4- s7
3- Leinfelder
KL. Porcelain esthetics for the 21st century. J Am Dent Assoc. 2000;131(suppl
1):s47- s51
4- McLean
J. The Science and Art of Dental Ceramics. Chicago, iL: Quintessence Publishing
Co inc; 1979
5- Piwowarczyk
A, Ottl P, Lauer HC, Kuretzky t. A clinical report and overview of scientifi
studies and clinical procedures conducted on 3M esPe Lava All-Ceramic system. J
Prosthodont. 2005;14(1):39-45.
6- elmaria
A, Goldstein G, Vijayaraghavan t, et al. An evaluation of wear when enamel is
opposed by various ceramic materials and gold. J Prosthet Dent.
2006;96(5):345-353.
7- Rama
Krishna Alla, Dental Materials Science, Jaypee Brothers Medical Publishers Pvt
Limited, New Delhi, India, 2013, 1st Edition, 333-354.
8- Saad
B.H. Farid, in Bioceramics: For Materials Science and Engineering, 2019
9- Kelly
JR, Denry I. Stabilized zirconia as a structural ceramic: an overview. Dental
materials. 2008 Mar 1;24(3):289-98.
10- Lughi
V, Sergo V. Low temperature degradation - aging - of zirconia: A critical
review of the relevant aspects in dentistry. Dental Materials.
2010;26(8):807–820.
11- Kenneth
JA . Dental Ceramics . In: Kenneth JA , Chiayi S , Ralph R , editors. Phillips
Science of dental materials . Elsevier ; 2013 . pp. 418 – 473
12- Al-Juaila
E, Osman E, Segaan L, Shrebaty M, Farghaly EA. Comparison of translucency for
different thicknesses of recent types of esthetic zirconia ceramics versus
conventional ceramics (in vitrostudy) Future Dental Journal. 2018
13- Tuncel
I, Turp I, Üşümez A. Evaluation of translucency of monolithic zirconia and
framework zirconia materials. J AdvProsthodont. 2016;8(3):181–186.
14- Ban S.
Reliability and properties of core materials for all-ceramic dental
restorations. Japanese Dental Science Review. 2008;44(1):3–21.
15- Stober
T, Bermejo JL, Rammelsberg P, Schmitter M. Enamel wear caused by monolithic
zirconia crowns after 6 months of clinical use. Journal of Oral Rehabilitation.
2014;41(4):314–322.
16- Kim MJ,
Oh SH, Kim JH, Ju SW, Seo DG, Jun SH, Ahn JS, Ryu JJ. Wear evaluation of the
human enamel opposing different Y-TZP dental ceramics and other porcelains.
Journal of Dentistry. 2012;40(11):979–988.
17- Bona
undefined, A undefined. D.; Pecho, O.E.; Alessandretti, R. Zirconia as a Dental
Biomaterial. Materials. 2015;8:4978–4991
18- Cavalcanti
AN, Foxton RM, Watson TF, Oliveira MT, Giannini M, Marchi GM. Y-TZP
Ceramics:Key Concepts for Clinical Application. Oper Dent. 2009;34(3):344–351
19- Russell
AG. Building Blocks. An overview of the various types of machinable blocks for
laboratory-based CAD/CAM systems. Inside Dental Technology. 2011;2(5)
20- Kedici
SP, Aksüt AA, Kílíçarslan MA, et al. Corrosion behaviour of dental metals and
alloys in different media. J Oral Rehabil 1998; 25: 800-8.
21- Sturzenegger
B, Feher A, Lüthy H, et al. Klinische Studie von Zirkonoxidbrücken im
Seitenzahngebiet hergestellt mit dem DCMSystem. Acta Med Dent Helv 2000; 5:
131-139.
22- Yildirim
M, Edelhoff D, Hanisch O, et al. Ceramic abutments--a new era in achieving
optimal esthetics in implant dentistry. Int J Periodont Restor Dent 2000; 20:
81-91.
23- Keith
O, Kusy RP, Whitley JQ. Zirconia brackets: an evaluation of morphology and
coefficients of friction. Am J Orthod Dentofacial Ortho 1994; 106: 605-14.










No comments:
Post a Comment